Chromium(VI), also known as hexavalent chromium or chromate, is a highly reactive and toxic oxidation state of the element chromium. It is one of the most dangerous and environmentally harmful forms of chromium, as it is highly toxic, carcinogenic and soluble in water. In industrial water and wastewater treatment, chromium(VI) is the focus of pollutant control, as it is used in various industries and its release into the environment is subject to strict legal regulations.
Table of contents
Technical background
Chromium(VI) is present in aqueous solutions in the form of chromate ions (CrO₄²-) or dichromate ions (Cr₂O₇²-), depending on the pH value of the water. These ions are highly mobile and strongly oxidizing, which increases their toxicity. In contrast to chromium(III), which is less toxic and usually precipitates as a solid hydroxide, chromium(VI) remains highly soluble in acidic and neutral environments and is therefore more difficult to remove.
Chromium(VI) is used in various industrial processes, including:
- Electroplating: Chromium(VI) is used in the surface finishing and chrome plating of metals to create a corrosion-resistant surface.
- Colors and pigments: Chromium(VI) is used in the production of pigments and colorants for special shades.
- Wood preservatives: Chromium(VI) is used in wood preservatives to protect the wood from insects and rot.
- Chemical industry: It is used as an oxidizing agent in chemical synthesis and for the production of chromium chemicals.
Toxicology and environmental pollution
Chromium(VI) is extremely toxic to humans and the environment. It can enter the human body through inhalation, ingestion or skin contact and is known to cause cancer, kidney damage, lung damage and allergic reactions. Because of these dangers, the use and disposal of waste water containing chromium(VI) is subject to strict environmental regulations.
- Mobility in the environment: Chromium(VI) ions are highly soluble in water and can easily enter groundwater, where they endanger drinking water resources. It is also highly toxic to aquatic organisms in water.
Wastewater treatment of chromium(VI)
Due to the high toxicity and mobility of chromium(VI), a thorough treatment of wastewater containing chromium(VI) is necessary before it can be discharged or reused. The most common method for treating chromium(VI) in industrial wastewater is chemical reduction to chromium(III), which can then be removed by precipitation as chromium hydroxide.
1. reduction of chromium(VI) to chromium(III)
The treatment of chromium(VI) usually begins with the reduction of hexavalent chromium to chromium(III), which is much less toxic and easier to remove. This is done by adding reducing agents such as:
- Sodium bisulfite (NaHSO₃)
- Sulphur dioxide (SO₂)
- Iron(II) sulphate (FeSO₄)
This reaction reduces the toxic chromium(VI) to chromium(III), which can then be precipitated in subsequent steps as insoluble chromium hydroxide [Cr(OH)₃]:
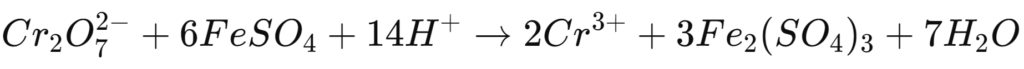
2. pH adjustment and precipitation
After reduction, the pH value of the wastewater must be raised to an alkaline range (pH 8-9) in order to precipitate the reduced chromium(III) as chromium hydroxide [Cr(OH)₃]. Here, the dissolved chromium(III) is converted into an insoluble form by adding sodium hydroxide (NaOH) or milk of lime (Ca(OH)₂), which can be removed by sedimentation or filtration can be separated by sedimentation or filtration.
3. flocculation and sedimentation
After precipitation, the chromium sludge formed is aggregated by flocculation with flocculants (such as polyelectrolytes) to enable faster sedimentation and better solid-liquid separation.

Photo: CP system ALMA CHEM MCW for the removal of heavy metals, AOX, hydrocarbons, cyanide and chromium (using sodium bisulphite)
4. ion exchange & activated carbon
In some cases, especially when very low chromium concentrations are required, additional steps such as activated carbon filtration or the use of ion exchangers are used to remove even the last traces of chromium from the water.

Photo: ALMA ION ion exchanger for the removal of specific pollutants such as chromium
Challenges in the treatment of chromium(VI)
Efficient reduction and pH control
Precise control of the reduction processes and the pH value is crucial to ensure that chromium(VI) is completely reduced to chromium(III). Incomplete reduction results in toxic chromium(VI) remaining in the wastewater.Sludge formation
The chemical precipitation of chromium leads to the formation of chromium sludge, which must be regularly dewatered and disposed of properly. This requires additional treatment and incurs disposal costs.Residual concentrations
Even after complete treatment, a residual concentration of chromium(VI) may remain in the water. In such cases, advanced processes such as ion exchange or membrane filtration must be used in order to comply with the legal limits.
Areas of application in which chromium(VI) is used
Chromium(VI) occurs in various branches of industry that require specific measures for the treatment of pollutants:
Electroplating industry
Chromium(VI) is used in electroplating baths to coat metals and create corrosion-resistant surfaces. The wastewater from these processes often contains high levels of chromium(VI), which must be treated before it is discharged or recycled.Leather and textile industry
Chromium(VI) is used in leather tanning to preserve and stabilize leather. In the textile industry, it is used for certain dyes. The waste water produced here must be thoroughly tested for chromium content and treated.Chemical and pharmaceutical industry
Chromium(VI) is used in the production of chemicals and as an oxidizing agent in the pharmaceutical industry. The treatment of this wastewater is often complex, as additional organic substances may be present that influence the reduction process.- Glass and ceramics industry
Chromium(VI) is used in glass coloring and ceramic production. Here too, waste water is produced that must be treated before being discharged into the environment.
Advantages of chromium(VI) treatment
Effective reduction to chromium(III): The reduction of chromium(VI) to chromium(III) is a proven method to drastically reduce the toxicity of wastewater and minimize environmental pollution.
Legal compliance: By fully treating chromium(VI), companies can ensure that they comply with strict legal regulations and minimize the risks to people and the environment.
Conclusion
Chromium(VI) poses a significant risk to the environment and human health and must therefore be comprehensively treated in industrial wastewater. The reduction of chromium(VI) to chromium(III), followed by precipitation and removal of the resulting chromium hydroxide, is a proven and effective method of reducing chromium contamination and complying with legal limits. Plants such as the ALMA CHEM MCW precipitation and flocculation plant rely on these technologies to support companies in the safe and environmentally friendly treatment of chromium.
For further information on our products, please feel free to contact us at any time!








