Nitrite is a chemical compound and an important intermediate in the nitrogen cycle. In wastewater and water treatment, nitrite plays a central role as an intermediate product in biological nitrogen conversion. It is formed during the oxidation of ammonium (NH₄⁺) to nitrate (NO₃-) in the nitrification process. At the same time, it is reduced back to gaseous nitrogen (N₂) during denitrification.
Table of contents
Technical background and development
In industrial and municipal wastewater treatment, nitrite typically occurs in two important biological processes:
1. nitrification:
- In the first step of nitrification, ammonium is oxidized to nitrite by ammonium-oxidizing bacteria (AOB), such as Nitrosomonas.
- Reaction equation:
- Nitrite is the intermediate product of this oxidation reaction and is further oxidized to nitrate in the second step by nitrite-oxidizing bacteria (NOB), such as Nitrobacter.
2. denitrification:
- In denitrification, nitrite, which is produced during nitrification or enters the wastewater stream due to chemical influences, is reduced to gaseous nitrogen by denitrifying bacteria under anoxic conditions.
- Reaction equation:
Importance of nitrite in water treatment
Nitrite is an unstable and potentially toxic substance that can have negative effects on water quality and the environment in high concentrations. It is less stable than nitrate and is toxic to aquatic life in high concentrations in biological systems. Especially in closed water circuits, e.g. in cooling water systems, nitrite can occur due to the reduction of nitrate or incomplete nitrification and cause corrosion problems.
Toxicity of nitrite
- Environmental hazard: Nitrite is toxic to aquatic organisms as it disrupts oxygen transport in the blood. In fish, a high nitrite concentration leads to the formation of methaemoglobin, which blocks oxygen transport and leads to oxygen deficiency.
- Health effects: Nitrite is also problematic in drinking water systems. If people ingest nitrite in large quantities, it can lead to methemoglobinemia, which impairs oxygen transport in the blood.
- Limits: For these reasons, there are strict legal limits for nitrite in drinking water and waste water. The limit value for nitrite in drinking water in the EU is 0.1 mg/l.

Photo: Denitrification and nitrification basin for industrial wastewater from the food industry in Chile, ALMA BHU BIO plant
Nitrite in industrial wastewater treatment
In industrial wastewater treatment, monitoring the nitrite content is crucial, as high nitrite concentrations can indicate incomplete nitrification or impaired denitrification. Particularly in industries with nitrogen-containing wastewater, such as the food and beverage industry, the chemical industry or the pharmaceutical industry, suitable measures must be taken to eliminate nitrogen in order to avoid nitrite.
Prevention and removal of nitrite
The avoidance or removal of nitrite can be achieved by various measures in wastewater treatment:
- Optimization of nitrification:
- Efficient nitrification, in which the ammonium is completely oxidized to nitrate, prevents the accumulation of nitrite. This requires a sufficient oxygen supply and stable operating conditions.
- Efficient denitrification:
- Complete denitrification, in which nitrite is quickly converted to nitrogen gas, prevents the accumulation of nitrite in anoxic zones.
- Monitoring and control:
- The nitrite content should be continuously monitored in order to quickly detect and rectify any malfunctions or imbalances in the biological process.
ALMAWATECH solutions for nitrite control
ALMAWATECH offers various system solutions for nitrogen removal and the prevention of nitrite accumulation in wastewater:
ALMA BHU Bio
Our biologically activated activated sludge plant combines nitrification and denitrification in a modular system designed for large wastewater flows. This plant can process up to 500 m³/h of wastewater with a high nitrogen load. Nitrification takes place in the aerated stage, which is regulated by automatic control systems to optimize oxygenation and other parameters.
ALMA BioFil Compact
This compact biofiltration system for smaller wastewater flows (up to 100 m³/h) combines nitrification and denitrification in one efficient system. It is ideal as pre-treatment for downstream membrane systems and reduces nitrogen compounds that could lead to fouling in reverse osmosis systems.
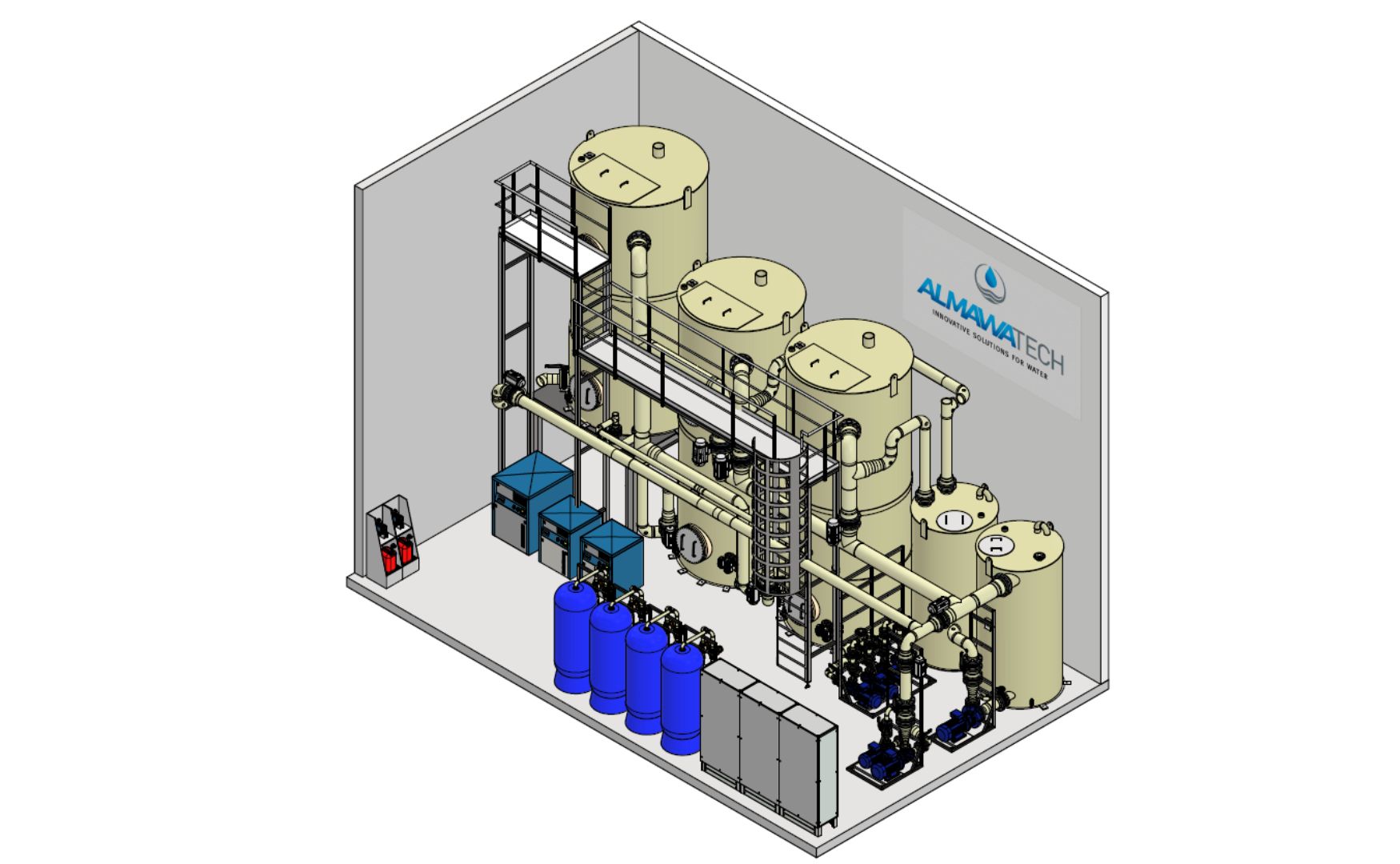
Photo: 3D design of our biologically activated ALMA BioFil Compact filtration system
ALMA BHU BAF
The large-scale concrete biofiltration plant offers efficient nitrification for wastewater flows of up to 1,000 m³/h. This plant uses an integrated system of aerobic and anoxic zones to maximize nitrogen removal. Particularly suitable for the food and beverage industry as well as municipal wastewater streams.

Photo: Photo of our biologically activated filtration, a combination process of mechanical cleaning and biodegradation(ALMA BHU BAF)
Conclusion
Nitrite is a critical intermediate in the nitrogen cycle that must be monitored and controlled in industrial and municipal wastewater treatment. Insufficient nitrification or denitrification can lead to an accumulation of nitrite, which can have a negative impact on both water quality and the health of aquatic organisms and humans. With ALMAWATECH 's tailor-made solutions, these risks can be minimized and efficient nitrogen removal ensured.








